Applying Human Factors Principles to Medical Device Labeling
Labeling is one of the first things a user encounters when interacting with a medical product; labeling can have a large impact on how a product is used and experienced. The focus of Human Factors/Usability Engineering is to understand how people interact with products and how user interface design affects these interactions. Human Factors/Usability Engineering considerations in the development of products involve the three major components of the device-user system: device users, device use environments, and device user interfaces. Device user interfaces are defined as all points of interaction between the user and the device. This includes all elements of the device with which the users can physically interact with, all sources of information transmitted by the device, including packaging and labeling, and all physical controls/display elements.
Learn more about evaluating Human Factors labeling and how Improvita can guide you through the process.
What is Considered Labeling?
Labeling is anything containing relevant information on the product or within the product’s packaging. Some of the most important labeling is on device labels, packaging labels, and instructions for use (IFU). The IFU has many areas where Human Factors Labeling principles can be applied so that the instructions are easy to follow. For example, making sure the print size is readable on the instructions, using a high contrast color between the words and background, and using visual aids to reduce the cognitive load for the user. The FDA has guidance documents for applying Human Factors principles to medical devices that include more details.
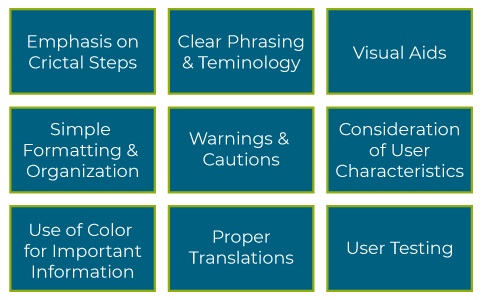
What are the Common Areas for IFU Improvement?
The instructions for how to use a medical device properly are critical to the success of the patient and healthcare provider. There are many areas within IFU designs that can be improved to help users understand critical information about the device. These include Human Factors Labeling principles such as emphasizing critical steps, color coding important information, and including visual aids.
Why are Human Factors Important in Medical Device Labeling?
Human Factors studies are important in labeling because they can provide insight into the thought process of potential users at an early stage in the product development process. Insights gathered during early testing can improve labeling and thus minimize risks associated with human errors. Improved labeling can help users make informed decisions about their health and improve their health by using the product correctly.
The FDA recommends that labeling, which is part of the user interface, be logical and intuitive to use. Human Factors principles can be utilized to create a well-designed user interface that will prevent the users from making decisions based on incorrect information that could lead to harm. The FDA requires the labeling to include specific warnings that pertain to the use of the product.
Label Comprehension Studies
The FDA can require a label comprehension (LC) study to be conducted for nonprescription drug products. Label comprehension studies are intended to evaluate the proposed product labeling for user comprehension and how the user makes decisions regarding the product according to their understanding of the labeling. The data from this study can identify areas on the label that would benefit from clearer or simpler presentation of important information.
Warning Labels
Warning labels contain critical information to reduce potential use errors and/or harm to the user. These warning labels often implement Human Factors principles such as bright colors, large text, or recognizable visuals to increase their likelihood of being acknowledged. All the risks associated with the warnings and cautions should be included in the risk management documents.
Why Choose Improvita for Human Factors Labeling Consulting?
At Improvita, we have extensive experience in medical device labeling and the evaluation of risks associated with the use of labeling. We can help guide your company through the Human Factors process to learn more about the current or future labeling of your product.
To learn more about Improvita’s Medical Device Labeling consulting services, contact us to discuss your specific needs.